
QOSCAT Universidad Oviedo
@qoscat_oviedo
👨🏼🔬👩🏻🔬 Group of Synthetic Organic Chemistry and Catalysis.
📍University of Oviedo @uniovi_info
⚗️ Organic Chemistry - Total synthesis - Photochemistry💡
ID: 1497907950819684359
27-02-2022 12:14:36
500 Tweet
503 Followers
514 Following


Check out our recent photoredox method in J Org Chem/Org Lett that enables the synthesis of 2-hydroxytrifluoroethylacetophenones from styrenes! Kudos to Gallego, Sarró and Ji for the good job! pubs.acs.org/doi/10.1021/ac… CatSyNanoMat Group Adela Vallribera

Best way to start holidays!! Our paper on generating axial chirality without atropisomers has been accepted in Angewandte Chemie. Another collaboration with our friend Elena Fernández Universitat Rovira i Virgili. Congrats to the true heroes Nerea and Josebe onlinelibrary.wiley.com/doi/10.1002/an…


Our latest publication in J. Am. Chem. Soc. demonstrates the use of amine-ligated boryl radical as XAT reagent in alkylation of amides using Cu-catalysis under oxidative conditions, circumventing the low nucleophilicity of amides RWTH Aachen pubs.acs.org/doi/10.1021/ja…

Discover @mechanochem for Organic and Inorganic Synthesis. Our latest review #editorschoice ACS Publications is out! Congrats F. Leon Javier Fernández pubs.acs.org/doi/full/10.10… Inorganic Chemistry J Org Chem/Org Lett Monash Chemistry Departamento de Química Orgánica e Inorgánica


Check our photoredox-catalyzed cyanation usign a bench-stable isocyanide Chemical Science pubs.rsc.org/en/content/art…. Congratulations to the team!!

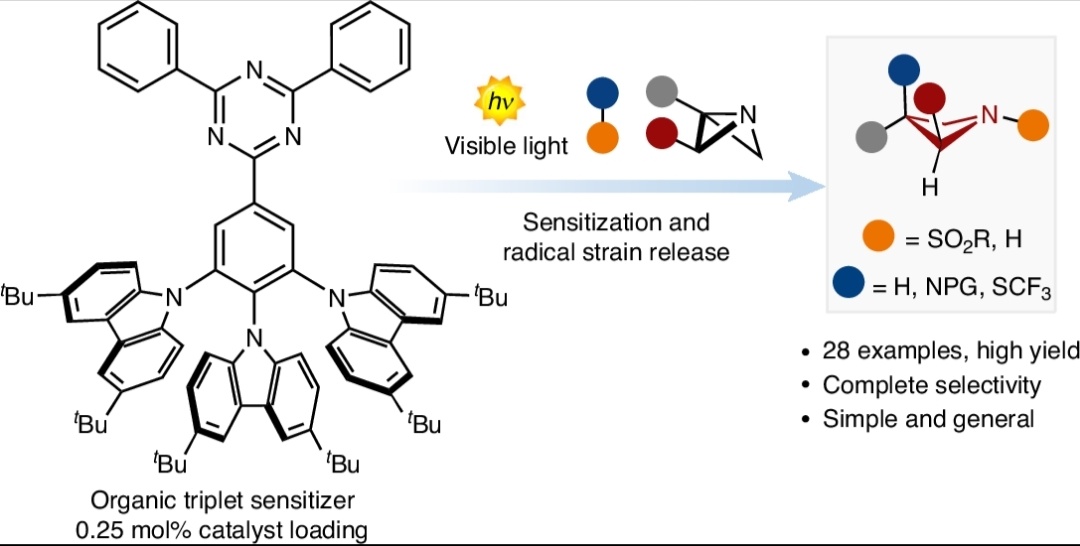
We're celebrating 🎉🎉 !! Our most recent work related to radical strain release photocatalysis for the synthesis of azetidines is now available Nature Catalysis in open access!!! 🔓🔓 A humongous job by all the team! 🐸nature.com/articles/s4192…🐸

A new chapter on the combination of gold species and enzymes has been disclosed in Adv. Synth. & Catal. Here, we demonstrate the compability of Ene-Reductases and gold catalyst in a cascade approach. Kudos to Lorena Escot for the hard work onlinelibrary.wiley.com/doi/10.1002/ad… BioorganicaUOVI

Fantastic collaborative work between QuimSinSos and our BioorganicaUOVI. Cover picture plus article in Adv. Synth. & Catal. merging the strengts of both Universidad Oviedo research groups. 👏👏 onlinelibrary.wiley.com/doi/10.1002/ad… onlinelibrary.wiley.com/doi/10.1002/ad…

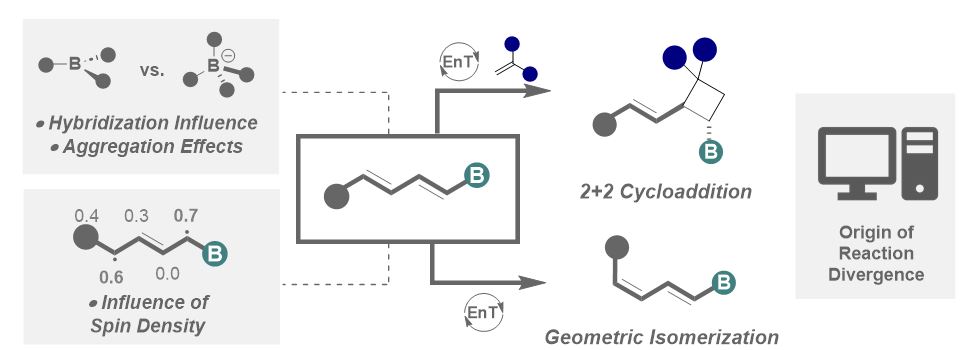
Delighted to share our recent collaborative effort with Ignacio Funes-Ardoiz "Regioselectivity of non-Symmetrical Borylated Dienes via EnT Catalysis: Unveiling the Relationship between Structure and Reactivity" posted on ChemRxiv Max Planck Institute of Colloids and Interfaces. Hope you enjoy! chemrxiv.org/engage/chemrxi…

Check out our last short review on C(sp³)-H functionalization via gold(I)-catalysis. Three kinds of reactivity are discussed. Thanks SYNTHESIS Journal Chemistry at Thieme. Finally, congratulations to all the authors. thieme-connect.com/products/ejour…


Just accepted, a Short Review on gold(I)-catalyzed C(sp3)-H bond funtionalization. Thanks to SYNTHESIS Journal and Chemistry at Thieme. Congratulations to Rubén Miguélez Omararto and Carlos Rodríguez thieme-connect.com/products/ejour…

BioLindlar Catalyst: Ene-Reductase-Promoted Selective Bioreduction of Cyanoalkynes to Give (Z)-Cyanoalkenes (Ivan Lavandera and co-workers) JorgeGlezRod Sergio González Granda Hirdesh Kumar Vicente Gotor Fernández UCL Chemistry Codexis Inc. Universidad Oviedo #openaccess onlinelibrary.wiley.com/doi/10.1002/an…

Our first work on electrophotocatalysis is finally published in OrgLett J Org Chem/Org Lett!! Congratulations, Bea Bea!! Jose Carlos Gonzalez Gomez pubs.acs.org/doi/10.1021/ac…


Mangish kept his eyes open in his copper-photo-electro project: no photo, no copper: electro-[2+2+2]-cyclization between alkynes (same or different ones) and nitriles. Congrats team, collaborators Rehbein Research Barham Lab; thanks to pec_idk; crc_325; doi.org/10.1002/anie.2…


nature.com/articles/s4158… Pleased to share our latest work in nature. This paper wraps up our research at ICIQ , and I’m grateful for their incredible support! Looking forward to continuing the journey with the Melchiorre Group Università di Bologna !

